With the rapid growth of the new energy industry, lithium iron phosphate batteries have become the new favorite of the market with their advantages such as high safety and long life. In production, Jet mill for Lithium Iron Phosphate play a key role in lithium iron phosphate preparation. As efficient ultrafine grinding equipment, they crush materials to micron or nanometer levels. This increases surface area and electrochemical performance. Jet mills grind sintered materials for uniform particle size distribution. This optimizes energy density, cycle life, and charge-discharge performance. The process is crucial for improving lithium iron phosphate quality and consistency.

Lithium Battery
A lithium battery consists of cathode, anode, separator, electrolyte, and casing. The cathode is a core material affecting energy density, safety, lifespan, and applications.
It accounts for 30-40% of material costs. The cathode is the largest and most valuable material in the battery industry.
According to material systems, cathode materials include lithium cobalt oxide, lithium manganese oxide, lithium iron phosphate, and ternary materials. Lithium iron phosphate is an olivine-structured cathode material. It is made from lithium, iron, phosphorus, and carbon sources through mixing, drying, sintering, and crushing.

Lithium Iron Phosphate
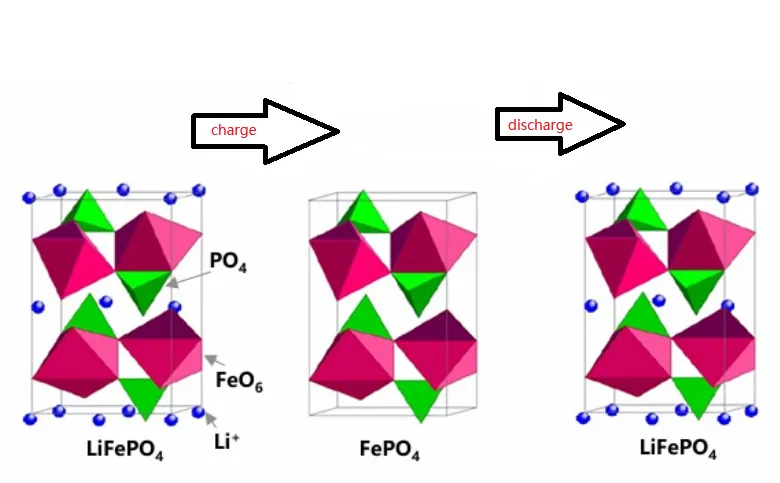
The molecular expression of lithium iron phosphate is LiFePO4. Its working principle in the charging and discharging process of lithium batteries is as follows:
When the lithium battery is charged, the lithium ion Li+ is separated from the lithium iron phosphate anode material LiFePO4, passes through the battery separator and electrolyte, and then embeds into the anode material, completing the charging process.
Important Factors Affecting the Performance of Lithium Iron Phosphate Cathode Material
Particle size
The particle size distribution of LiFePO₄ crystals greatly affects cathode material rate performance.
Under the same conditions, smaller particles shorten Li⁺ transport paths. Smaller particle size improves rate performance and enables faster charging and discharging.
Specific capacity
The specific capacity of LiFePO₄ significantly affects the battery’s gravimetric energy density. Under the same conditions, higher specific capacity increases energy density. Higher specific capacity means greater battery capacity for the same mass.
Compacted density
The compaction density of LiFePO₄ significantly affects the battery’s volumetric energy density. Under the same conditions, higher compaction density increases volumetric energy density. Higher compaction density means greater battery capacity for the same volume.
Specific surface area
The specific surface area of LiFePO₄ greatly affects rate and low-temperature performance. Under the same conditions, a larger surface area increases contact with the electrolyte. Better conductivity improves rate performance, enabling faster charging and discharging.
Impurity content
The impurity content of LiFePO₄ affects the battery’s electrochemical performance and safety. Impurities include calcium, sodium, copper, chromium, and zinc. Excessive impurities increase self-discharge and shorten battery lifespan. High impurity levels raise the risk of separator damage, reducing battery safety.
Moisture content
The moisture content of LiFePO₄ affects battery electrochemical performance, safety, and lifespan. Excess moisture reacts with the electrolyte, forming gas and hydrofluoric acid. This causes battery swelling, corrosion, and reduced safety and performance.
Other indicators
Particle shape, tap density, carbon content, pH and other electrochemical properties of lithium iron phosphate.
Preparation method of lithium iron phosphate
There are various methods for LiFePO₄ preparation. Based on material reaction states, they are classified into solid-phase and liquid-phase synthesis methods. According to different preparation methods, the preparation process of lithium iron phosphate is different, and the corresponding equipment is also different. Jet mill for Lithium Iron Phosphate are inseparable.
Solid phase method-Carbothermal reduction method
Raw Material Refinement Pre-treatment: For the solid-phase method, iron source (e.g., FePO₄), lithium source (e.g., Li₂CO₃), and carbon source (e.g., glucose) are thoroughly mixed. The jet mill, through high-speed airflow-generated shear force and collision, crushes raw materials to micron or sub-micron levels. This significantly improves particle fineness and dispersion uniformity, preventing local reaction inconsistency or composition segregation due to coarse particles.
Particle Size Control and Classification: The jet mill with air classifier offers precise particle size classification. It can control the D50 (median particle size) and distribution range of the final product. This optimizes lithium ion diffusion paths and electronic conductivity during subsequent sintering, enhancing material compaction density and rate performance.
Liquid phase method – self-evaporation liquid phase synthesis method
Pre-treatment and Homogenization of Precursors
Refinement of Solid Raw Materials: Although the liquid-phase method primarily involves solution reactions, some processes require pre-crushing solid raw materials such as lithium source (e.g., LiOH) and iron source (e.g., FePO₄·2H₂O) to micron-sized particles. This improves their dissolution rate and dispersion in the solvent. The jet mill, through high-speed airflow shear forces, efficiently crushes raw materials to sub-micron levels, reducing particle agglomeration and ensuring uniformity in subsequent liquid-phase reactions.
Secondary crushing and classification of dried particles
In the self-evaporation liquid-phase method, the lithium iron phosphate precursor often forms wet particles through solution evaporation and crystallization. After drying, issues like agglomeration or uneven particle size distribution may occur. The jet mill can perform secondary grinding on the dried coarse particles, breaking apart agglomerates and producing monodispersed, micron-sized particles.
The jet mill’s classification system can select particles within a specific size range (e.g., D50 = 1-3 μm). This prevents issues such as decreased compaction density from overly fine particles or increased ion diffusion resistance from overly coarse particles, optimizing the material’s electrochemical performance (e.g., rate capacity and cycle life).

Conclusion
The Jet mill for Lithium Iron significantly enhances the quality of lithium iron phosphate. It ensures uniform particle size and improves electrochemical performance. By optimizing particle size and dispersion, the jet mill boosts rate capacity and cycle life. This process plays a crucial role in advancing lithium iron phosphate battery technology.
Epic powder
Epic Powder, 20+ years of work experience in the ultrafine powder industry. Actively promote the future development of ultra-fine powder, focusing on crushing,grinding,classifying and modification process of ultra-fine powder. Contact us for a free consultation and customized solutions! Our expert team is dedicated to providing high-quality products and services to maximize the value of your powder processing. Epic Powder—Your Trusted Powder Processing Expert!
